Unveiling the elliptical nature of hydrogen bonds
– Kritika Gaur
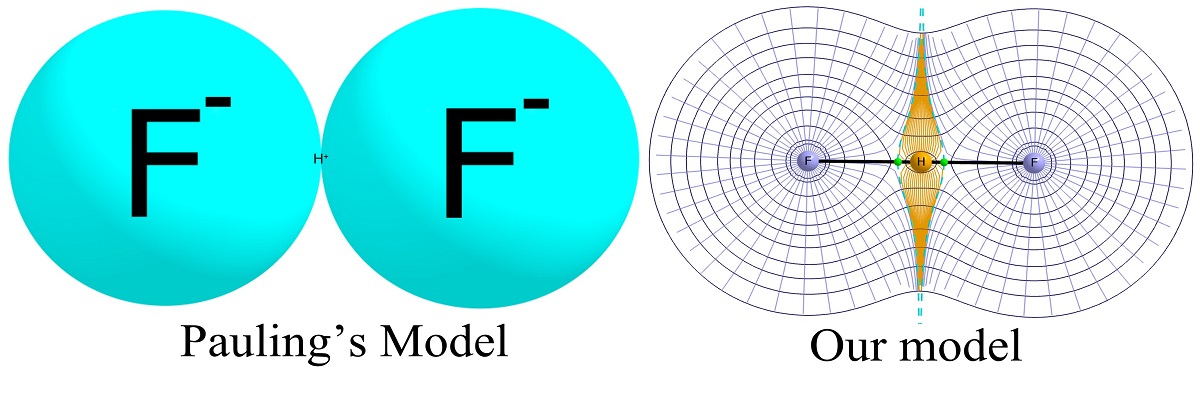
Biflouride anion (FHF)- is considered the strongest hydrogen bond. Linus Pauling’s book suggested the left-side structure. Shahi and Arunan used Bader’s Atoms in Molecules theory to show H occupies a significant space and that its shape is anisotropic (Image: Abhishek Shahi)
Hydrogen bonding is a key noncovalent interaction that plays a central role in phenomena such as protein folding and molecular organisation.
In a recent study, Elangannan Arunan and his team from the Department of Inorganic and Physical Chemistry (IPC), IISc, undertook a systematic and chemically informed approach to understand the radii of H-atom in hydrogen bonds. The group adopted the Quantum Theory of Atoms in Molecules (QTAIM) analysis to quantitatively demonstrate the elliptical nature of hydrogen atom in hydrogen bond complexes, earlier assumed to have spherical symmetry. They introduced an electron density-based definition of hydrogen bond radii.
Traditionally, such bonds have been described using van der Waals (vdW) radii (which represents the closest distance at which one atom can approach another) of the hydrogen donor (D) and acceptor (A) atoms. In a typical D–H—A interaction (H-atom in molecule D interacting with molecule A), if the distance between the atoms D and A is shorter than the sum of their individual vdW radii, the interaction is classified as a hydrogen bond. But this geometric criterion inherently assumes that the atoms are spherical.
Employing a rigorous electron density-based framework, the new study suggests that hydrogen atoms exhibit pronounced anisotropy (a direction-dependent property) in their electron density distribution. This ellipticity is characterised in both isolated D–H molecules and D–H—A complexes. The study further reveals that in non-linear hydrogen bonds, the HBR (Hydrogen Bond Radii) exhibits dependence on the D–H—A angle.
Importantly, the researchers show that the anisotropy parameter (g) of the H-atom has correlates with the electronegativity of the D-atom, the hybridisation state of D-atom, and the strength of H-bond with the acceptor. Particularly, weak hydrogen bond acceptors possess less ellipticity in comparison to strong hydrogen bond acceptors.
These findings underscore the sensitivity of hydrogen’s electron density to its chemical environment. Such a theoretical framework could also be extended to estimate the radii for other non-covalent bonds such as halogen bond, chalcogen bond, and so on in the future.
REFERENCE:
Shahi A, Arunan E, The Long and Short Radii for Hydrogen in Hydrogen Bonded Complexes, Journal of Computational Chemistry (2026).
https://onlinelibrary.wiley.com/doi/epdf/10.1002/jcc.70313
LAB WEBSITE: https://ipc.iisc.ac.in/~ea/




