One of the first clear signs of the onset of Alzheimer’s disease is the loss of short-term memory, the ability to remember recent events. However, scientists believe that changes in the brain begin decades before clinicians notice such signs.
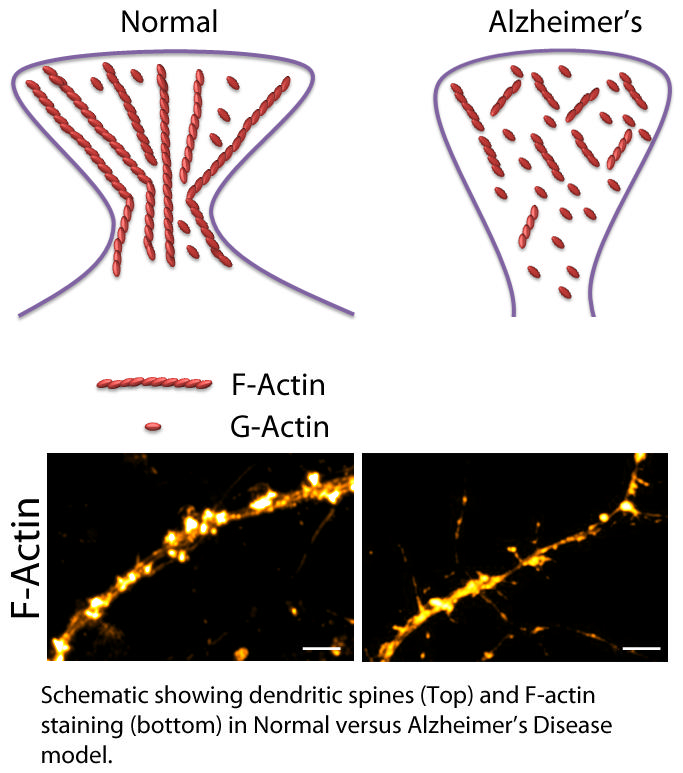
Researchers at the Centre for Neuroscience and Centre for Brain Research at the Indian Institute of Science (IISc) have now identified a key protein in the brain that is broken down early on in Alzheimer’s disease, affecting communication between nerve cells that is important for memory formation. The protein, called F-actin (fibrillar actin, a polymer of the protein G-actin), is responsible for maintaining the shape of mushroom-shaped projections called dendritic spines on the surface of a nerve cell. These spines protrude into synapses, the junctions between nerve cells, and act as docking spots for other neurons to connect and transmit signals.
The researchers found for the first time that F-actin is broken down very early on in Alzheimer’s disease, affecting the shape and number of spines, which in turn disrupts synaptic communication and leads to memory deficits.
Alzheimer’s disease is thought to affect 60-70% of the 50 million people worldwide who suffer from some form of dementia. There is currently no cure or treatment to slow down its progression. Identifying key molecules involved in the initial stages may help detect it early and develop drugs that delay its onset or slow it down, allowing patients to lead a better quality of life.
Research has shown that one of the key early events is the disruption of synapses caused by loss of, or defects in dendritic spines, which interrupts the flow of information between nerve cells. However, what causes this disruption has remained unclear.
In this study, the researchers used mice with genetically introduced genes that are mutated in familial Alzheimer’s disease (AD mice) to look at proteins involved in maintaining dendritic spine shape and number. Inside these spines, proteins called G-actins assemble to form long filaments called F-actins that control the spine’s structure. The researchers found that in AD mice as young as 1 month old, the F-actin/G-actin balance was disrupted, with more F-actin being broken down into G-actin, leading to misshapen and lost spines. By contrast, the formation of toxic protein clumps called amyloid plaques, which is one of the first clinical symptoms, is typically seen only at 7-8 months of age in AD mice.
To test if F-actin loss had any effect on behaviour, the researchers used a paradigm called contextual fear conditioning. In these experiments, mice are trained to fear a specific location or context by giving them a mild electric shock. When normal mice are placed in the same arena the next day, they freeze, remembering and anticipating the shock. AD mice as young as 2 months old, however, did not freeze as expected, indicating that they had forgotten the context.
Remarkably, when the researchers injected a chemical into AD mice that prevented F-actin from breaking down, they found that the mice were able to regain their normal fear response. “That was very striking. When we stabilized the F-actin, we were able to see the behaviour recovery,” says Reddy Kommaddi, first author and DBT Ramalingaswami Fellow at the Centre for Neuroscience.
The researchers also tested normal mice with a chemical that causes F-actin to breakdown, and found that they seemed to forget their fear, just like AD mice. “We deliberately depolymerized F-actin in normal mice and showed that this itself is enough for the deficit to manifest,” says Smitha Karunakaran, another author and scientist at the Centre for Brain Research.
To find out if similar effects were also happening in humans, the researchers looked at post-mortem brain tissue samples of patients with Alzheimer’s disease, who had been studied for more than a decade before their death. The samples were obtained from collaborator David Bennett at the Rush Alzheimer’s Disease Center in Chicago, USA. Just like the AD mice, these samples also showed a gradual breakdown of F-actin over time, as their symptoms — memory loss and accumulation of plaques — worsened.
Taken together, these results indicate a direct link between F-actin loss and early behavioural changes that lead to Alzheimer’s disease, the researchers say.
“Because F-actin is a structural protein, it gives shape to all cells in the body, and is present everywhere. It could potentially become a biomarker,” says Vijayalakshmi Ravindranath, senior author and Professor at the Centre for Neuroscience.
The researchers are currently working on understanding what causes F-actin to break down. They found, for example, that nerve cells treated with a toxic protein called amyloid beta 42 — which eventually gives rise to the classical AD plaques — show reduced F-actin levels. The team is currently working on unravelling the molecular mechanisms involved.
Website: http://www.jneurosci.org/content/38/5/1085


